Brent white created date. 2 the oxidation number of sulfur is 2.
Inspirational Periodic Table Oxidation Numbers Pdf Tablepriodic Priodic Tablepriodicsample Periodic Table Periodic Table Words Periodic Table With Names
Microsoft word 14 04 oxidation numbers worksheet doc author.

Oxidation numbers worksheet pdf. This exercise is designed to teach the student how to assign oxidation numbers. 3 2 the polyatomic anion has a charge of 2. When you are ready you will be given an examination over this material.
Sio 2 si o 3. The sum of the oxidation numbers in a polyatomic ion is equal to the charge on the ion. Ptcl 4 2 n.
Let s examine the oxidation numbers of some common elements. The oxidation number of an element in a monatomic ion equals the charge of the ion. Cl 2 cl 16.
Oxidation number exercise answers page 57 oxidation number exercise do not hand in this work sheet. Rules 1 the oxidation number of the atoms in any free uncombined element is zero 2 the sum of the oxidation numbers of all atoms in a compound is zero 3 the sum of the oxidation numbers of all atoms in an ion is equal to the charge of the ion 4 the oxidation number of fluorine in all its compounds is 1. Ptcl 6 2 c.
Calculating oxidation numbers for sulfur. Compute the number of electrons lost in the oxidation and gained in the reduction from the o n. A pure element has an oxidation number of 0.
Worksheet assigning oxidation numbers name period oxidation number rules. Na 2 o 2 na o 2. Multiply one or both of these numbers by appropriate.
7 15 2005 10 38 28 pm. Rules for assigning oxidation numbers the oxidation number of any uncombined element is 0 the oxidation number of a monatomic ion equals the charge on the ion. Formula element and oxidation number formula element and oxidation number 1.
The sum of the oxidation number of all the elements in a compound equals 0. Al 2o 3 f. Notice the periodic trend among the main group.
Sbf 6 i. Microsoft word 14 04 oxidation numbers worksheet doc author. Brent white created date.
A monoatomic ion has an oxidation number equal to its charge. Complete the examination by yourself and hand it in to receive credit. For example the oxidation number of the oxygen in the oxide ion o 2 is 2.
From the changes in o n identify the oxidized and reduced species 3. The oxidation number of fluorine in a compound is always 1. We can begin by recalling that the charge on an ion corresponds to the sum of the oxidation numbers.
Give oxidation numbers for the underlined atoms in these molecules and ions. Assign oxidation numbers to all elements in the reaction 2. Oxidation numbers worksheet directions.
Oxidation number method 1. We assign oxygen an oxidation number of 2 and write the equation c in so. Use the rules for assigning oxidation numbers to determine the oxidation number assigned to each element in each of the given chemical formulas.
Mno 4 m. H 3aso 3 h. The more electronegative element in a binary compound is assigned the number equal to the charge it would have if it were an ion.

Redox Oxidation Numbers Practice Worksheet By The Scientific Classroom Practices Worksheets Chemistry Lessons Worksheets

37 Simple Balancing Equations Worksheet Ideas Https Bacamajalah Com 37 Simple Balancing Equation Chemistry Worksheets Chemical Equation Balancing Equations

Electron Configuration Worksheet Answers Part A Worksheets For Electron Configuration Chemistry Worksheets Word Problem Worksheets

Oxidation Numbers Sulphur Exhibits Oxidation Numbers Of 2 0 2 4 And 6 High School Chemistry Chemistry Chemistry 101

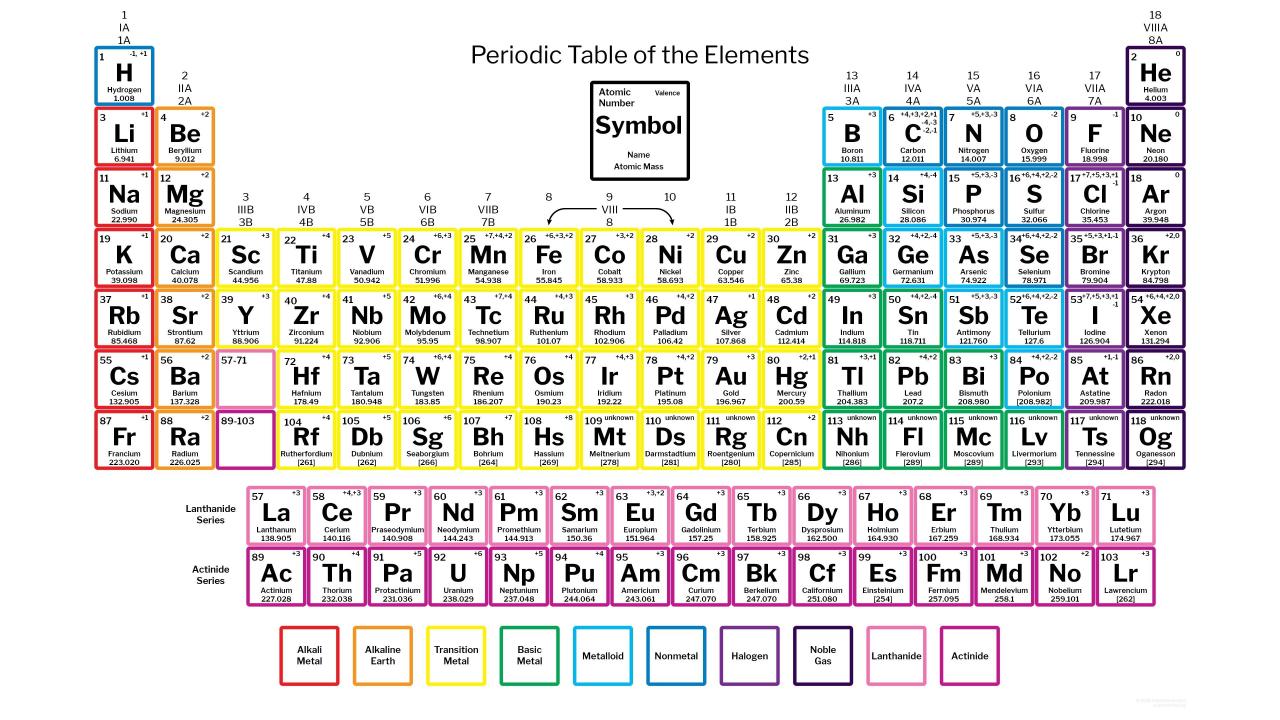
Oxidation Number Periodic Table Periodic Table Printable Periodic Table Chart Periodic Table Of The Elements

New Periodic Table Grade 7 Pdf Printable Math Worksheets Multiplication Printable Math Worksheets Periodic Table

Students Will Be Applying The Assigning Oxidation Numbers Rules They Learned In Class To A Practic Practices Worksheets Chemistry Lessons Flirting Quotes Funny

Redox Reactions Redox Reactions Chemistry Education Chemistry Worksheets

7 Readable Language Arts Worksheets 7th Grade In 2020 Chemistry Worksheets Science Notes Element Chart

Oxidation Reduction Worksheet Answers Free Read Oxidation Reduction Reaction Worksh In 2020 Scientific Notation Word Problems Worksheet Template Pre Algebra Worksheets

Oxidation Reduction Worksheet Answers Worksheet 25 Oxidation Reduction Reactions In 2020 Scientific Notation Word Problems Word Problem Worksheets Factoring Quadratics

Picture Redox Reactions Free Math Worksheets Chemistry Notes

50 Periodic Table Puzzle Worksheet Answers In 2020 With Images Persuasive Writing Prompts Worksheet Template Worksheets

Oxidation Number Chart Pinning This To Print Later Ugh Chemistry Teaching Chemistry Chemistry Number Chart

Oxidation Reduction Worksheet Answers 25 Redox Review Worksheet Answers In 2020 Scientific Notation Word Problems Word Problem Worksheets Pre Algebra Worksheets

Oxidation States Of Transition Metals Chemistry Libretexts Oxidation State Transition Metal Electron Configuration

Oxidation Numbers Worksheets Practice Questions Hs Chemistry Number Worksheets Chemistry This Or That Questions



