Also includes adding subtracting multiplying and dividing fractions. Practice basic multiplication skills with these printable worksheets and games.

This Worksheet Includes 15 Practice With Factoring Trinomials As Well As Special Cases Such As Difference Factoring Polynomials Polynomials Writing Equations
Page 3 lesson 1.

Factoring practice worksheet. M f2 q0p1 m2v kktu xtja 0 nsroyf8t dw6anr ce l bljl gcg 0 1 ea qltl n fr eirg lh7t 8s7 frgezsxerrmvbende. Factoring trinomials of the form 2 where 1 pg. Here is a graphic preview for all of the factors worksheets you can select different variables to customize these factors worksheets for your needs.
F f wmkajd zeb owfiytuhd oidnufxi fn dijt 1e i 2acl cg neub sroag m2y u worksheet by kuta software llc kuta software infinite algebra 2 name factoring quadratic expressions date period. This bundle is perf. Included here are factoring worksheets to factorize linear expressions quadratic expressions monomials binomials and polynomials using a variety of methods like grouping synthetic division and box method.
15 review more practice factoring with pizzazz worksheets pg. The factors worksheets are randomly created and will never repeat so you have an endless supply of quality factors worksheets to use in the classroom or at home. Algebra multiplying binomials and factoring practice riddle worksheet mini bundle this is a set of three of my best selling riddle practice worksheets.
Practice factoring prime numbers worksheets 192784 prime factorization worksheets answers 192785 prime numbers worksheet year 4 luxury number patterns new math. Simplifying fractions equivalent fractions operations with fractions. Greatest common factor gcf find the gcf of the numbers.
S h2w0k1l2 n skluet oay ps qo7f 5tmw8a5r0er altlkce n i haelelq 1r eiogshit ys d 6r gedszejr vvrepds g a fm 6a gd ge3 ow9ihthm kimn9f 5imn0iotre o favl bg sezb nrkam y1f v worksheet by kuta software llc kuta software infinite algebra 1 name factoring trinomials a 1 date period. They are designed to practice and reinforce the concept of multiplying binomials factoring a 1 and factoring when a 1. Factors worksheets printable factors and multiples worksheets.
Factoring using the greatest common factor factor each expression by factoring out the gcf. These factoring polynomials worksheet pdfs with answer keys offer high school students immense practice.

Factoring Polynomials Gcf Factoring Polynomials Algebra Polynomials

Factoring Over Real Numbers Precalculus Real Numbers Polynomials

Factoring Polynomials Puzzle From Just Mathematics On Teachersnotebook Com 2 Pages School Algebra Factoring Polynomials Math

More Factoring Over Real Numbers Polynomials Worksheets Factoring Polynomials

Search And Shade Worksheets Hoppe Ninja Math Teacher Blog High School Algebra School Algebra Algebra Worksheets
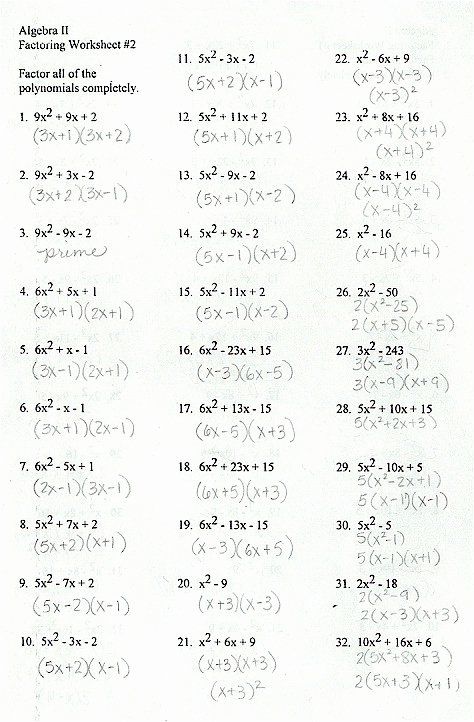
Factoring Trinomials Worksheet Answers Luxury Factoring General Trinomials In 2020 Factoring Polynomials Factor Trinomials Mathematics Worksheets

Factoring Trinomials Worksheet Answers Awesome 10 Best Of Factoring Polynomials Practice Worksheet Chessm In 2020 Factoring Polynomials Factor Trinomials Polynomials

Lego Batman Factoring Polynomials Search Amp Shade Factoring Polynomials Math Teacher Polynomials

Factoring Practice Worksheet In 2020 Algebra Worksheets Factoring Quadratics Quadratics

Pin By Jamie Riggs Missmathdork On Teachers Pay Teachers Missmathdork Algebra Worksheets Factor Trinomials Learning Mathematics

Factoring Polynomials Trinomials Activity Beginner Factoring Trinomials Activity Factoring Polynomials Education Math

St Patrick S Day Theme Factoring Polynomials Matching Activity Students Will Practice Factoring Polyno Factoring Polynomials Polynomials Factoring Quadratics

Algebra 1 Worksheets Monomials And Polynomials Worksheets Factoring Polynomials Algebra Worksheets Polynomials

Factoring Polynomials Matching Polynomials Factoring Polynomials Algebra Interactive Notebooks

Factoring Trinomials Puzzle Activity Freebie Factoring Quadratics Activities Factoring Quadratics Factoring Trinomials Activity

Solving Polynomial Equations Worksheet Answers Best Of Solving Polynomial Equations By Factoring Worksheet T In 2020 Algebra Worksheets Factoring Quadratics Quadratics

Factoring Polynomials Worksheet Answers Fresh Factoring Trinomials Worksheet Easy By Elizabeth Gra In 2020 Factoring Polynomials Polynomials Character Trait Worksheets

Algebra 2 Mixed Factoring Practice Worksheet Practices Worksheets Solving Quadratic Equations Algebra Worksheets