These are the strongest of the intermolecular forces. Intermolecular force worksheet key.

Pin By Megan Cozort On Chem In 2020 Intermolecular Force Hydrogen Bond Chemistry
A pf3 dipole dipole force b h2co dipole dipole force c hf hydrogen bonding.

Intermolecular forces worksheet key. Weakest intermolecular force that results from the constant motion of electrons. Intermolecular forces the tendency of a substance to be found in one state or the other under certain conditions is largely a result of the forces of attraction that exist between the particles comprising it. Intermolecular forces displaying top 8 worksheets found for this concept.
You may find it useful to draw lewis structures to find your answer. Intermolecular forces worksheet answers are on page 3 4. Predict the molecular shape of each of the following.
Intermolecular force worksheet 2 key identify the strongest intermolecular force present in pure samples of the following substances. Identify the strongest intermolecular force present in pure samples of the following substances. Hf hydrogen bonding forces molecules that have hydrogen attached to an o n or f can form hydrogen bonds.
You may find it useful to draw lewis structures to find your answer. 2 co c hf 2 explain how dipole dipole forces cause molecules to be attracted to one another. Intermolecular forces worksheet 2 answers 1 using your knowledge of molecular structure identify the main intermolecular force in the following compounds.
List all types of imfs that would occur in each of the following you should have a good. Intermolecular forces and boiling points. We will concentrate on the forces between molecules in molecular substances which are called intermolecular forces.
Some of the worksheets for this concept are intermolecular forces work work 15 intermolecular forces work chemistry 20 intermolecular forces work chem1101 work 7 intermolecular forces information work intermolecular forces intramolecular between types of intermolecular forces intermolecular forces. What is the strongest intermolecular force present for each of the following molecules. So2 like water so2 is a bent molecule h2o ch2cl2.
1 hydrogen h 2 london dispersion forces 2 carbon monoxide co london dispersion forces 3 silicon tetrafluoride sif 4 london dispersion forces 4 nitrogen tribromide nbr 3 dipole dipole forces 5 water h 2 o hydrogen bonding 6 acetone ch 2. Dipole dipole forces the strongest intermolecular force in a polar molecule that cannot form hydrogen bonds is the dipole dipole force e. Occurs in all molecules title microsoft word 5 20a 20b molecular geometry and forces wkst key doc.
Do the problems on your own before looking at the answers. 1 using your knowledge of molecular structure identify the main intermolecular force in the following compounds.

Intermolecular Forces Cmap Intermolecular Force Force Physics Chemistry

4 Types Of Intermolecular Forces Google Search In 2020 Intermolecular Force Covalent Bonding Molecules
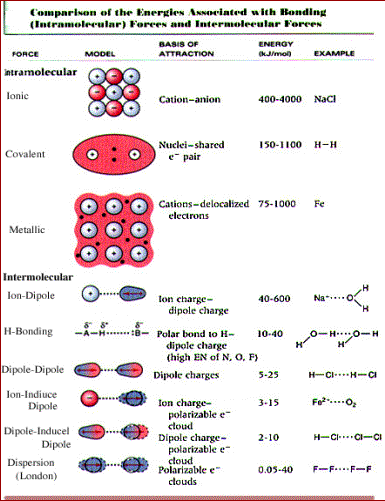
505 Stmatterimf Htm Chemistry Education Intermolecular Force Teaching Chemistry

Pin By Kate Scott On Chemistry Chemistry Worksheets Science Worksheets Chemistry Classroom

Pin On Worksheet Templates For Student

Learn Quiz On Kinetic Interpretation Of Temperature Chemistry Quiz 262 To Practice Free Chemistry Mcqs Que College Chemistry Chemistry This Or That Questions

Intermolecular And Intramolecular Forces Mr Lowe S Science And Chemistry Chemistry Education Chemistry Teaching Chemistry

Chemistry Notes Liquids Solids And Intermolecular Forces Chemistry Notes Intermolecular Force Organic Chemistry

Chemie Notizen Arten Von Chemischen Reaktionen Chemistry Notes Arten Chemie Chemischen Chemistry Notes Notizen Quimica Quimica Organica Fisica

Liquids Solids And Intermolecular Forces Intermolecular Force Chemistry Notes Chemistry

Intermolecular Forces Worksheet Mazes 3 Levels Distance Learning In 2020 Intermolecular Force Force Ap Chemistry

Vsepr Theory 3 Levels Of Mazes Vsepr Theory Kids Worksheets Printables Radical Expressions

Intermolecular Forces And Hydrogen Bonding Review Question Trail Intermolecular Force This Or That Questions Hydrogen Bond

Msextavourscienceclasses Intermolecular Forces Nastas Lena Intermolecular Force Ap Chem Chemistry

Chemistry Notes Liquids Solids And Intermolecular Forces In 2020 Chemistry Notes Intermolecular Force Chemistry

How To Predicting Boiling Point And Melting Point Trends Chemistry Classroom Intermolecular Force Teaching Chemistry

Pin On Worksheet Templates For Student

Ap Chemistry Core Concept Cheat Sheet 12 Atomic Structures Key Chemistry Terms Boxes And Arrow Chemistry Worksheets College Chemistry Chemistry Education


