Compute the number of electrons lost in the oxidation and gained in the reduction from the o n. Multiply one or both of these numbers by appropriate.

Grades 10 12 Redox Balancing Redox Reactions Practice Worksheet In This Hands On Worksheet Students Will Be R Redox Reactions Reactions Practices Worksheets
Redox reactions answer key determine the oxidation number of the elements in each of the following compounds.

Balancing redox reactions worksheet 1 answer key. What is the oxidation number of carbon in nahco3. From the changes in o n identify the oxidized and reduced species 3. H 2 co 3 h.
Ws 5 balancing redox reactions in acid and basic solution. If you are struggling as well then all you need balancing equations worksheet with answers. Balance each redox equation.
Identify the species being oxidized and reduced in each of the. If you get stuck try asking another group for help. Split reaction into half reactions reduction and oxidation.
And that is wrong because there is an electron in the final answer. 4e 4h o 2 2h 2 o. 2o 2 2f 2 o 2 4f 2.
In the reaction 2k cl2 2kcl the species. Balancing redox equations method 1. You should try to answer the questions without referring to your textbook.
Redox practice worksheet name. Often students lose hope and struggle to solve it. Oxidation number method 1.
I aq no 2 aq i 2 s no g i. Balancing requires a lot of practice knowledge of reactions formulae valances symbols and techniques. In which substance is the oxidation number of nitrogen zero.
Use the half reaction method. Balancing redox reactions worksheet 1 answer key the following redox reaction takes place in acidic solution. Balancing redox reactions worksheet 1 balance each redox reaction in.
You can in a half reaction but remember half reactions do not occur alone they occur in reduction oxidation pairs 2 here are the correct half reactions. P cu 2 æ cu h2po4 ph3 i2 æ h3po2 i no2 æ no3 no. Assign oxidation numbers to all elements in the reaction 2.
Assume all are spontaneous. Overall scheme for the half reaction method. Mno4 c2o4 2 æ mno2 co2.
In the reaction al0 cr3 al3 cr0 the reducing agent is a. Clo2 æ clo2 clo3. You cannot have electrons appear in the final answer of a redox reaction.
Mn 2 bio3 æ mno4 bi 3 mno4 s2o3 2 æ s4o6 2 mn 2 clo3 cl æ cl2 clo2. Fe 3 o 4 fe. The half equation method separates the oxidation and reduction of a redox reaction in half reactions.
Balance the reaction and indicate which reactant is oxidized and which reactant is being reduced.

Picture Redox Reactions Free Math Worksheets Chemistry Notes
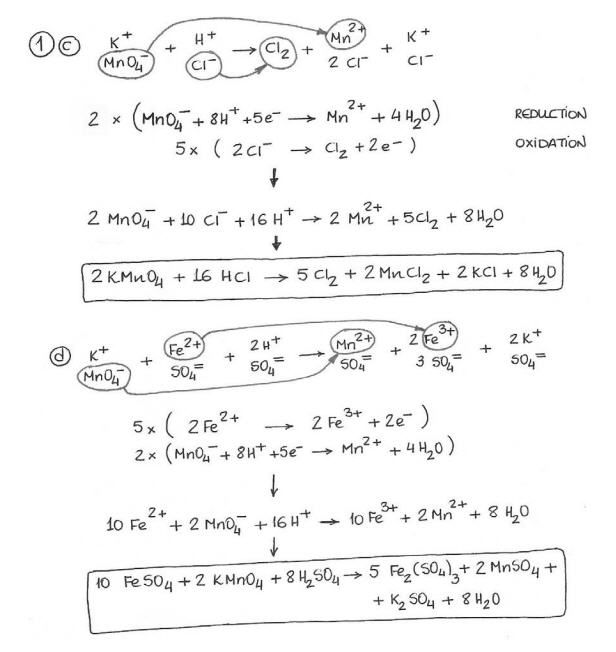
Pin By Sarah Brogan On Balancing Of Chemical Reaction Equations Distance Time Graphs Worksheets Redox Reactions

Tang 02 Balancing Redox Reactions 2 Redox Reactions Organic Reactions Reactions

Balance A Redox Reaction Basic Solution Redox Reactions Ap Chemistry Chemistry

Balancing Chemical Equations Worksheets With Answers Chemical Equation Balancing Equations Chemistry Worksheets

Balancing Chemical Equations Worksheets With Answers In 2020 Balancing Equations Chemical Equation Equations

Balancing Chemical Equations Worksheet Chemical Equation Chemistry Worksheets Balancing Equations

Types Of Chemical Reactions Worksheet 19 Sample Balancing Chemical Equations Wor In 2020 Chemistry Worksheets Sight Word Worksheets Kindergarten Subtraction Worksheets

Download Balancing Equations 48 Chemical Equation Equations Balancing Equations

Chemistry Multiple Multiple Choice Types Of Chemical Reactions Chemistry Chemical Reactions Multiple Choice

Tang 02 Balancing Redox Reactions Redox Reactions Teaching Chemistry Teaching Science

Balancing Chemical Equations Worksheet Balancing Equations Literal Equations Chemical Equation

Redox Reactions Exercise With Solutions Redox Reactions Chemical Equation Reactions

37 Simple Balancing Equations Worksheet Ideas Https Bacamajalah Com 37 Simple Balancing Equation Chemistry Worksheets Chemical Equation Balancing Equations

Tang 02 Balancing Redox Reactions 2 Lezioni Di Scienze Chimica Scienza

Balancing Chemical Equations Chemical Equation Balancing Equations Chemistry Worksheets

Tang 02 Balancing Redox Reactions 2 Redox Reactions Chemistry Lessons Chemistry Education

Pin By Luci D Amico On Balancing Of Chemical Reaction Redox Reactions Chemistry Worksheets Chemistry Lessons

Classifying Chemical Reactions Worksheet Answers New 16 Best Of Types Chemical Reactions Worksheets In 2020 Chemistry Worksheets Chemical Reactions Teaching Chemistry


